CYSTIC FIBROSIS
Cystic fibrosis also known as CF or mucoviscidosis is an autosomal recessive genetic disorder that affects most critically the lungs, and also the pancreas, liver, and intestine. It is characterized by abnormal transport of chloride and sodium across an epithelium, leading to thick, viscous secretions.The name cystic fibrosis refers to the characteristic scarring (fibrosis) and cyst formation within the pancreas. Difficulty breathing is the most serious symptom and results from frequent lung infections that are treated with antibiotics and other medications. Other symptoms, including sinus infections, poor growth, and infertility affect other parts of the body. CF is caused by a mutation in the gene for the protein cystic fibrosis transmembrane conductance regulator (CFTR). This protein is required to regulate the components of sweat, digestive fluids, and mucus.
CFTR
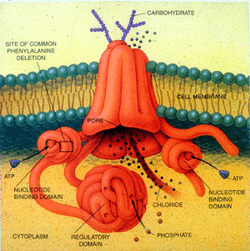
A CFTR protein molecule
Cystic fibrosis transmembrane conductance regulator (CFTR) is a protein that in humans is encoded by the CFTR gene. CFTR is an ABC transporter-class ion channel that transports chloride and thiocyanate ions across epithelial cell membranes.
Structure:- The CFTR gene is approximately 189 kb in length. CFTR is a glycoprotein with 1480 amino acids. The protein consists of five domains. There are two transmembrane domains, each with six spans of alpha helices. These are each connected to a nucleotide binding domain (NBD) in the cytoplasm. The first NBD is connected to the second transmembrane domain by a regulatory "R" domain that is a unique feature of CFTR, not present in other ABC transporters. The ion channel only opens when its R-domain has been phosphorylated by PKA and ATP is bound at the NBDs.The carboxyl terminal of the protein is anchored to the cytoskeleton by a PDZ-interacting domain.
Function: - CFTR functions as a -activated ATP- gated anion channel, increasing the conductance for certain anions (e.g. Cl–) to flow down their electrochemical gradient. ATP-driven conformational changes in CFTR open and close a gate to allow transmembrane flow of anions down their electrochemical gradient. This in contrast to other ABC proteins, in which ATP-driven conformational changes fuel uphill substrate transport across cellular membranes. Essentially, CFTR is an ion channel that evolved as a 'broken' ABC transporter that leaks when in open conformation. The CFTR is found in the epithelial cells of many organs including the ling ,liver, pancreas, digestive tract, reproductive tract, and skin. Normally, the protein moves chloride and thiocyanate ions (with a negative charge) out of an epithelial cell to the covering mucus. Positively charged sodium ions follow these anions out of the cell to maintain electrical balance. This increases the total electrolyte concentration in the mucus, resulting in the movement of water out of cell by osmosis. In epithelial cells with motile cilia lining the bronchus and the oviduct, CFTR is located on cell membrane but not on cilia. In contrast to CFTR, ENaC is located along the entire length of the cilia. These findings contradict a previous hypothesis that CFTR normally down regulates ENaC by direct interaction and that in CF patients, CFTR cannot down regulate ENaC causing hyper-absorption in the lungs and recurrent lung infections. In sweat glands, CFTR defects result in reduced transport of sodium chloride and sodium thiocyanate in the re absorptive duct and saltier sweat. This was the basis of a clinically important sweat test for cystic fibrosis before genetic screening was available
Structure:- The CFTR gene is approximately 189 kb in length. CFTR is a glycoprotein with 1480 amino acids. The protein consists of five domains. There are two transmembrane domains, each with six spans of alpha helices. These are each connected to a nucleotide binding domain (NBD) in the cytoplasm. The first NBD is connected to the second transmembrane domain by a regulatory "R" domain that is a unique feature of CFTR, not present in other ABC transporters. The ion channel only opens when its R-domain has been phosphorylated by PKA and ATP is bound at the NBDs.The carboxyl terminal of the protein is anchored to the cytoskeleton by a PDZ-interacting domain.
Function: - CFTR functions as a -activated ATP- gated anion channel, increasing the conductance for certain anions (e.g. Cl–) to flow down their electrochemical gradient. ATP-driven conformational changes in CFTR open and close a gate to allow transmembrane flow of anions down their electrochemical gradient. This in contrast to other ABC proteins, in which ATP-driven conformational changes fuel uphill substrate transport across cellular membranes. Essentially, CFTR is an ion channel that evolved as a 'broken' ABC transporter that leaks when in open conformation. The CFTR is found in the epithelial cells of many organs including the ling ,liver, pancreas, digestive tract, reproductive tract, and skin. Normally, the protein moves chloride and thiocyanate ions (with a negative charge) out of an epithelial cell to the covering mucus. Positively charged sodium ions follow these anions out of the cell to maintain electrical balance. This increases the total electrolyte concentration in the mucus, resulting in the movement of water out of cell by osmosis. In epithelial cells with motile cilia lining the bronchus and the oviduct, CFTR is located on cell membrane but not on cilia. In contrast to CFTR, ENaC is located along the entire length of the cilia. These findings contradict a previous hypothesis that CFTR normally down regulates ENaC by direct interaction and that in CF patients, CFTR cannot down regulate ENaC causing hyper-absorption in the lungs and recurrent lung infections. In sweat glands, CFTR defects result in reduced transport of sodium chloride and sodium thiocyanate in the re absorptive duct and saltier sweat. This was the basis of a clinically important sweat test for cystic fibrosis before genetic screening was available
Mutation
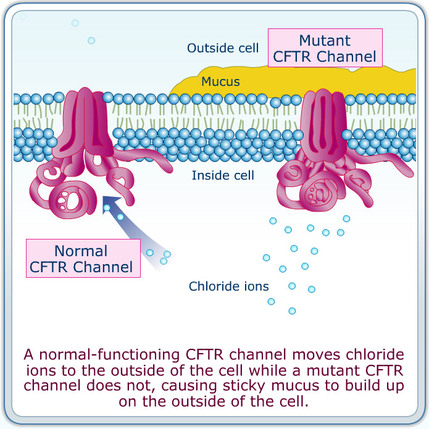
Comparision between a normal CFTR and an Mutant CFTR.
Well over one thousand mutations have been described that can affect the CFTR gene. Such mutations can cause two genetic disorders, congenital bilateral absence of vas deferens and the more widely known disorder cystic fibrosis. Both disorders arise from the blockage of the movement of ions and, therefore, water into and out of cells. In congenital bilateral absence of vas deferens, the protein may be still functional but not at normal efficiency, this leads to the production of thick mucus, which blocks the developing vas deferens. In people with mutations giving rise to cystic fibrosis, the blockage in ion transport occurs in epithelial cells that line the passageways of the lungs, pancreas, and other organs. This leads to chronic dysfunction, disability, and a reduced life expectancy. The most common mutation, ΔF508 results from a deletion (Δ) of three nucleotides which results in a loss of the amino acid phenylalanine (F) at the 508th position on the protein. As a result the protein does not fold normally and is more quickly degraded. The vast majority of mutations are quite rare. The distribution and frequency of mutations varies among different populations which has implications for genetic screening and counseling. Mutations consist of replacements, duplications, deletions or shortenings in the CFTR gene. This may result in proteins that may not function, work less effectively, are more quickly degraded, or are present in inadequate numbers. It has been hypothesized that mutations in the CFTR gene may confer a selective advantage to heterozygous individuals. Cells expressing a mutant form of the CFTR protein are resistant to invasion by the Salmonella typhi bacterium, the agent of typhoid fever, and mice carrying a single copy of mutant CFTR are resistant to diarrhea caused by cholera toxin.

